
Recent Posts
Child Health, News
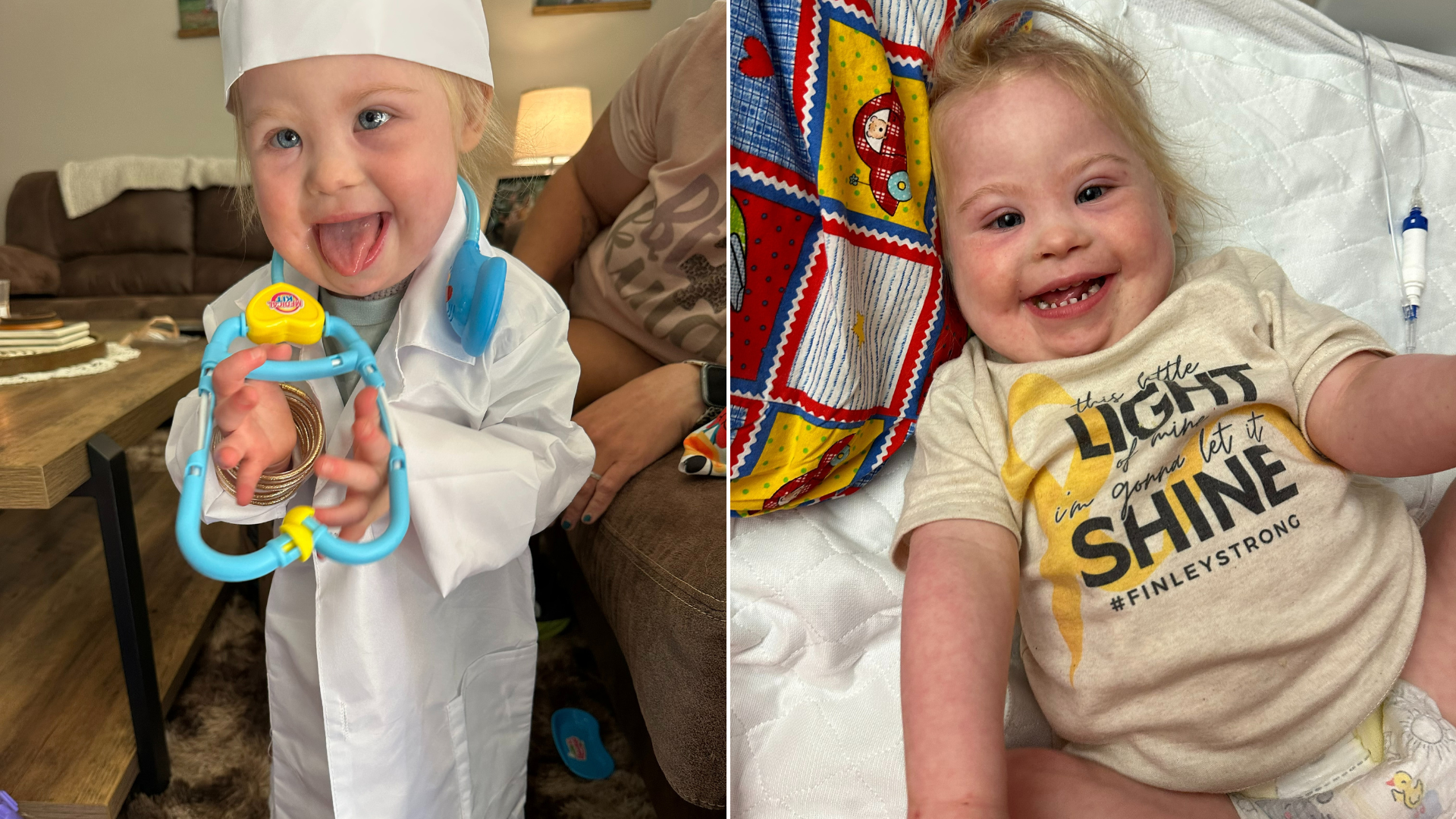
Against all odds: Toddler survives stroke, brain swelling and heart arrhythmia, thanks to care at Levine Children’s

Primary Care, News
Spring-Clean Your Health

News, Child Health
Bringing Home Baby: A Parents’ Guide to Making Things Safe and Sound

News
What Does it Mean When You Receive a Cancer Diagnosis?

News, Child Health
Empowering Children: How to Teach Them about Boundaries and Unwanted Touch

News, Child Health
Game On: Safeguarding Young Athletes from Concussions and Injuries

News, Women's Health
Atrium Health’s Female Cardiologists Lead the Way in Improving Women’s Heart Health

News, Child Health
Under pressure: Helping your child or teen cope with school-related stress
